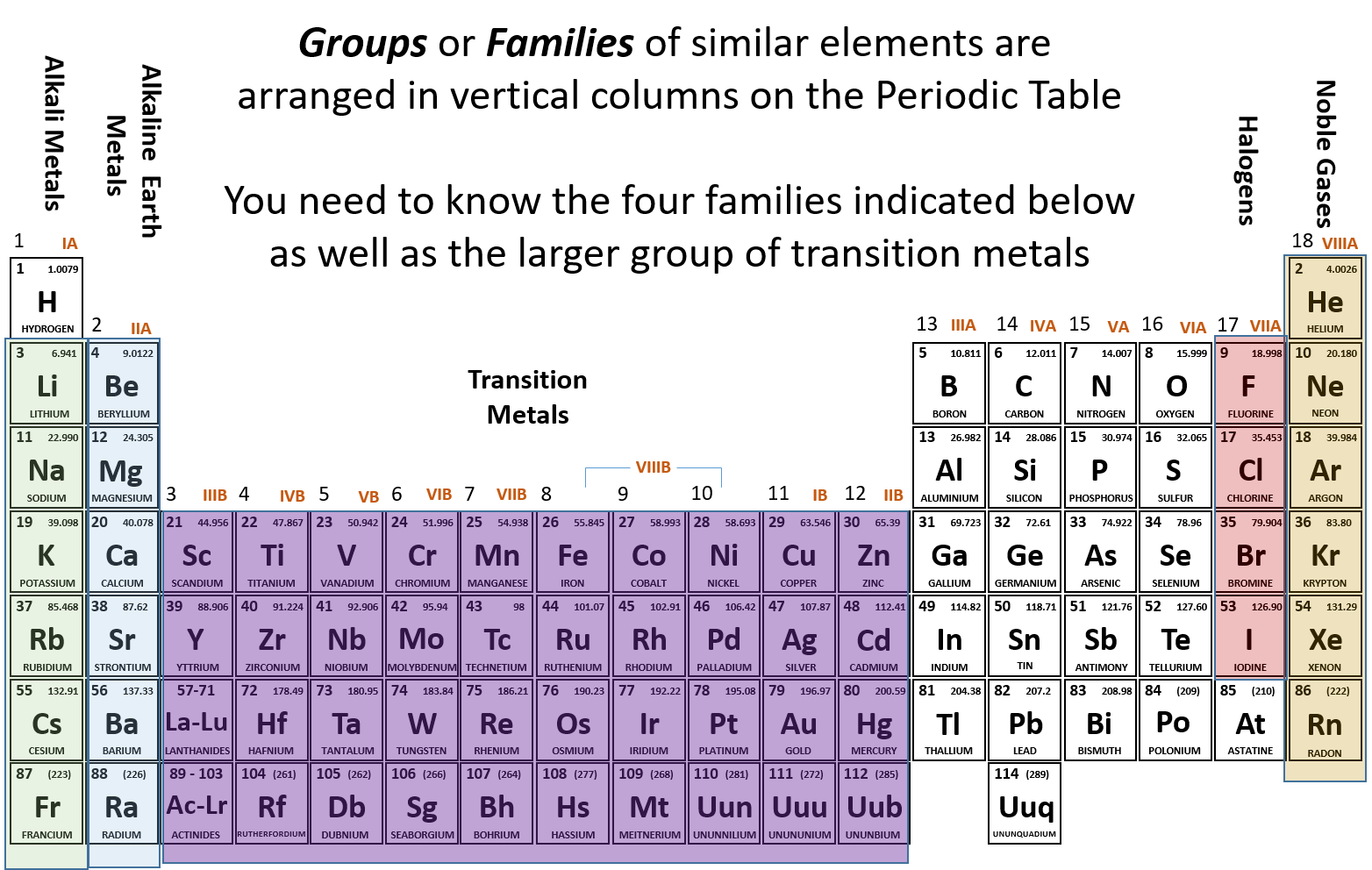
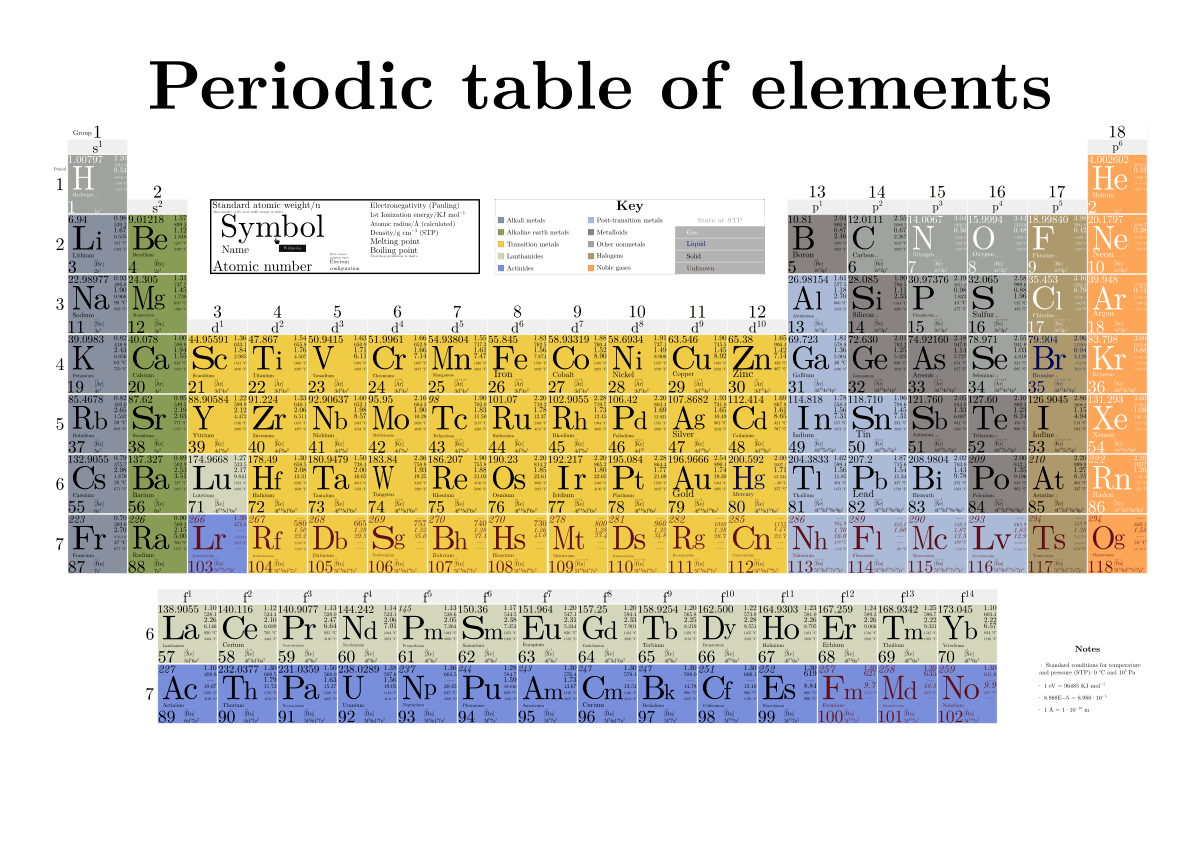
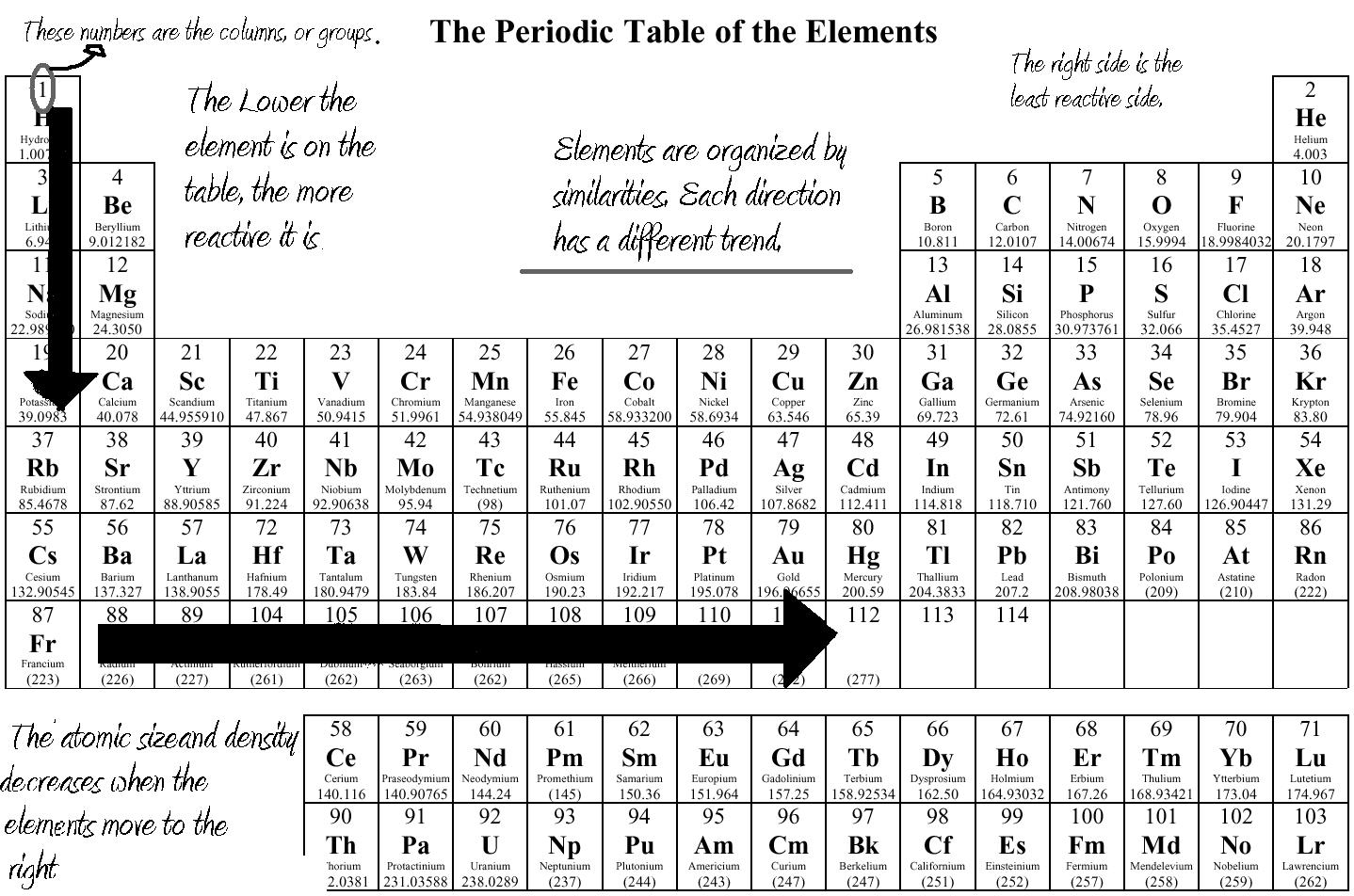
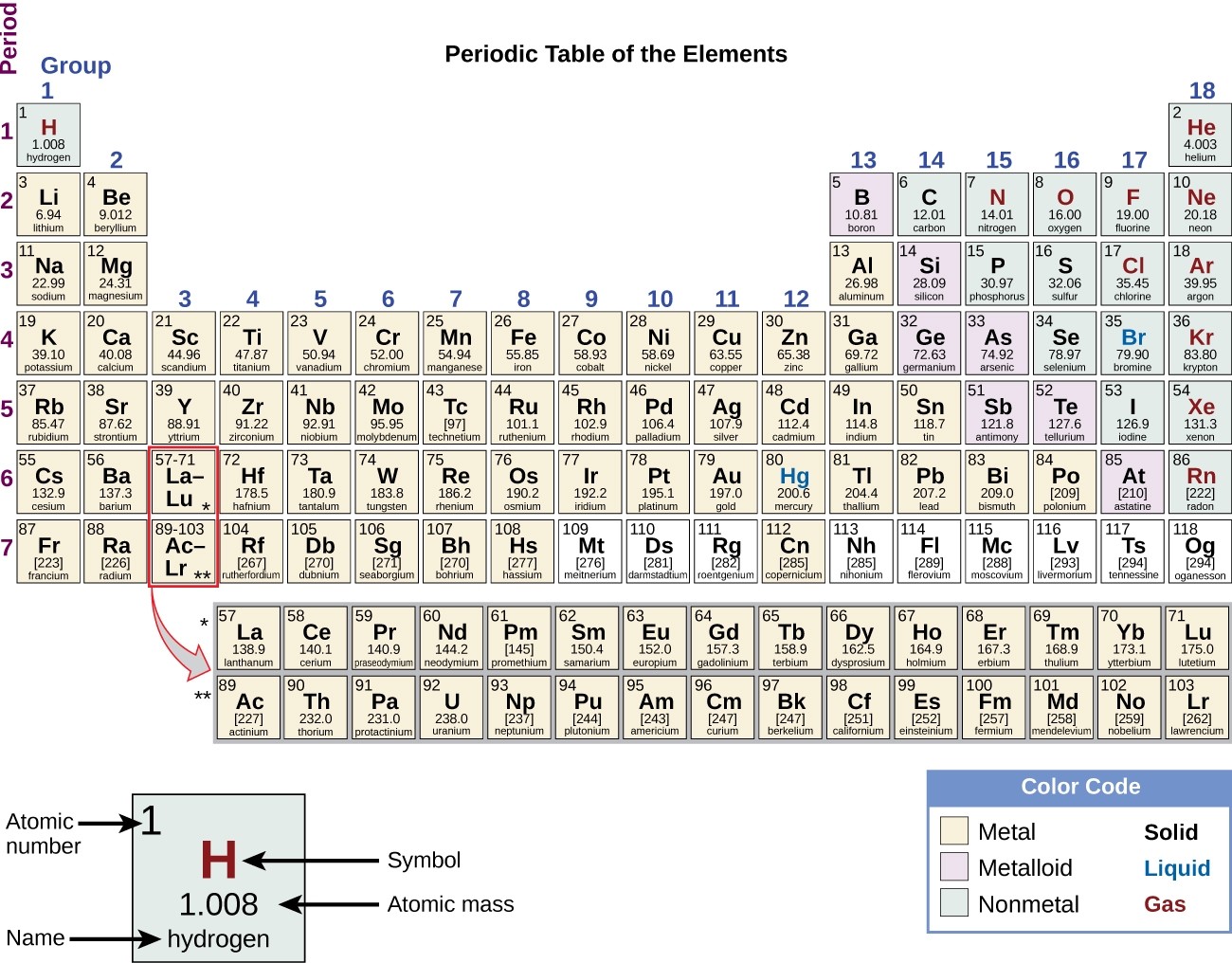
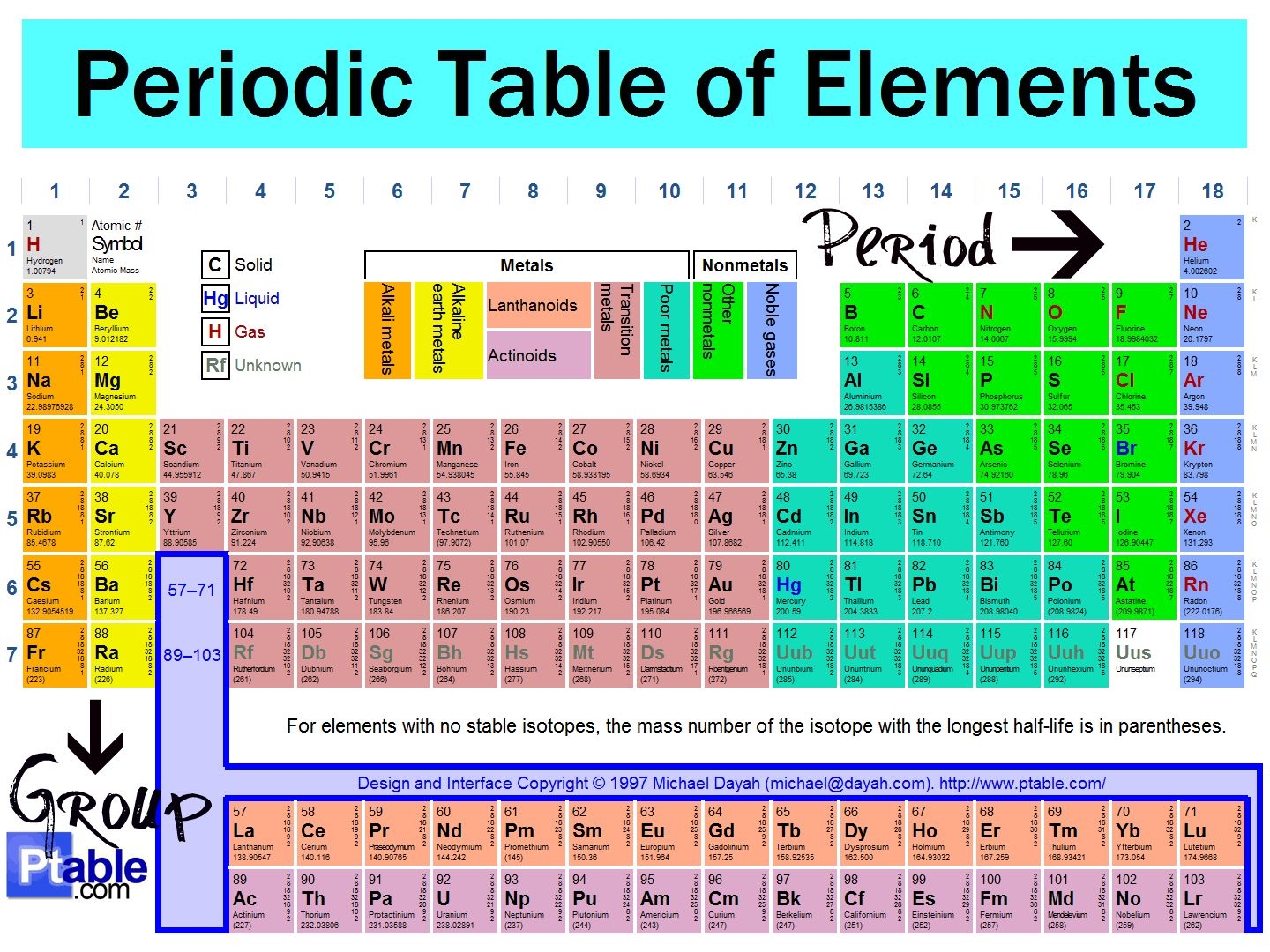
Have you ever wondered about the vertical column on the periodic chart? It’s known as a group or family, and each group has similar chemical properties.
Elements within a group have the same number of valence electrons, making them behave similarly in chemical reactions. This is why elements in the same group often exhibit similar characteristics.
Vertical Column On The Periodic Chart
Vertical Column On The Periodic Chart
For example, Group 1 elements like sodium and potassium are highly reactive metals that readily lose their outermost electron to form a +1 ion. This reactivity is due to their single valence electron.
On the other hand, Group 18 elements like helium and neon are noble gases that have a full outer electron shell, making them stable and unreactive. This is why noble gases are often used in lighting, lasers, and other applications.
Understanding the vertical columns on the periodic chart can help us predict the chemical behavior of elements and how they will react with other substances. It’s fascinating to see how patterns emerge in the properties of elements within the same group.
Next time you look at the periodic chart, take a closer look at the vertical columns and see if you can spot any similarities between elements within the same group. It’s like uncovering a hidden code that helps us make sense of the vast world of chemistry!
How To Read The Periodic Table Overview U0026 Components Expii
Discuss The Main Feature Of Groups In The Long Form Of Periodic Table
Trends On The Periodic Table Science Blog
The Periodic Table Atoms First OpenStax
1 2 Elements Make Up The Periodic Table Yumna s Science Blog